iLink™ corneal cross-linking is a minimally invasive outpatient procedure that combines the use of ultraviolet light and specially formulated eye drops to stiffen and strengthen corneas that have been weakened by disease or refractive surgery. Cross-linking is considered the standard of care around the world for progressive keratoconus and corneal ectasia following refractive surgery.2
Is iLink™ Covered by Insurance?
The medical necessity of iLink™ has become widely recognized. As a result, commercial insurance coverage for the procedure is now over 95% in the United States.
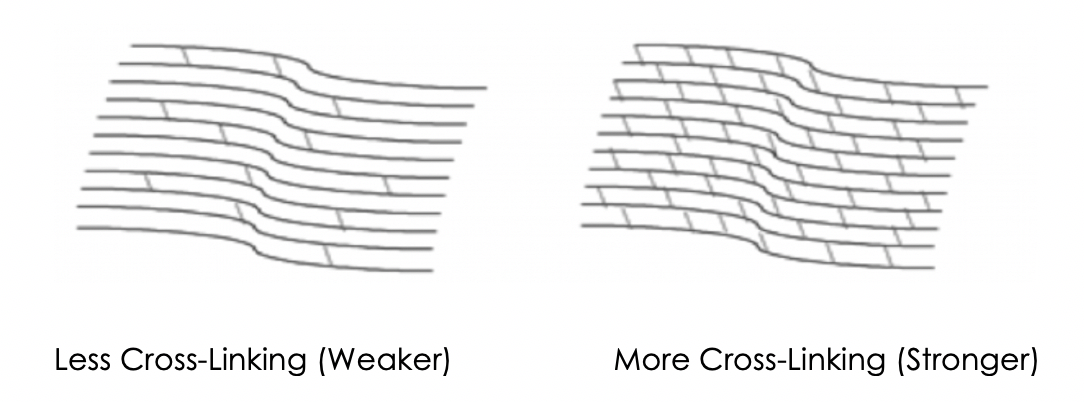
Corneal Cross-Linking3
- Creates new corneal collagen cross-links
- Results in a shortening and thickening of the collagen fibrils
- Leads to the stiffening of the cornea
Riboflavin
Under the conditions used for iLink™ corneal cross-linking, specially formulated pharmaceutical-strength riboflavin eye drops called Photrexa® (riboflavin 5’-phosphate ophthalmic solution) and Photrexa® Viscous (riboflavin 5’-phosphate in 20% dextran ophthalmic solution) help enable the cross-linking reaction.
Ultraviolet Light (UV)
iLink™ corneal cross-linking applies an artificial source of ultraviolet light from a machine called the KXL System once the cornea has been soaked in the Photrexa® and Photrexa® Viscous eye drops. This process works to stiffen the cornea by increasing the number of molecular bonds, or cross-links, in the collagen.
Combining Riboflavin and UV Light
Using Photrexa® and Photrexa® Viscous riboflavin eye drops, combined with ultraviolet light from the KXL system, the iLink™ procedure stiffens and strengthens the cornea to slow or halt progressive keratoconus.
Is Cross-Linking Right for Me?
Patients who have been diagnosed with progressive keratoconus or corneal ectasia following refractive surgery should ask their doctor about iLink™ corneal cross-linking.
Our practice is proud to offer patients the first and only FDA-approved therapeutic solution for the treatment of progressive keratoconus. Now, patients who once had little to no therapeutic option to treat keratoconus have the opportunity to slow or halt the progression of this sight-threatening disease.
For more information about the iLink™ procedure for the treatment of keratoconus and corneal ectasia following refractive surgery, visit livingwithkeratoconus.com.
Frequently Asked Questions
Click here to access the FAQs about iLink™ corneal cross-linking.
2. Gomes JAP, Tan D, Rapuano CJ, et al. Global consensus on keratoconus and ectatic diseases. Cornea. 2015;34(4):359-369.
3. Beshwati IM, O’Donnell C, Radhakrishnan H. Biomechanical properties of corneal tissue after ultraviolet-A-riboflavin crosslinking. J Cataract Refract Surg. 2013;39(3):451-462.





